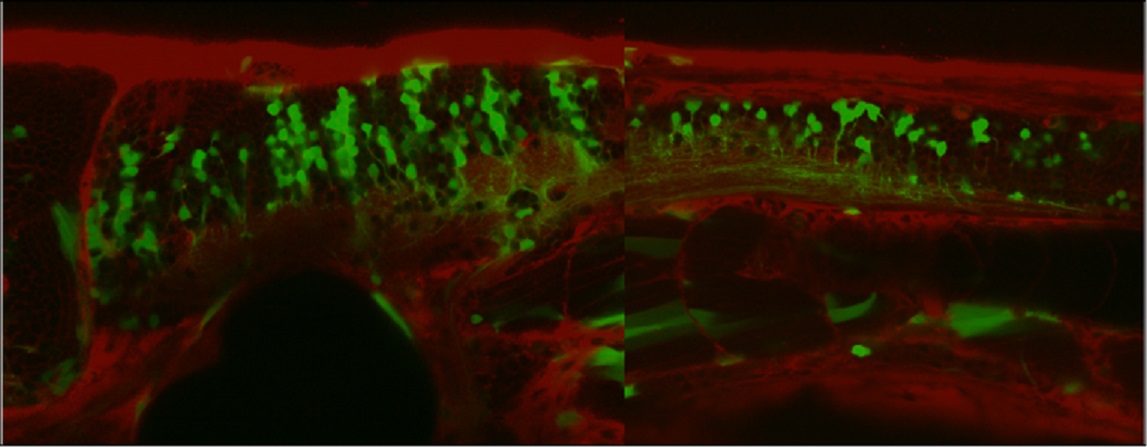
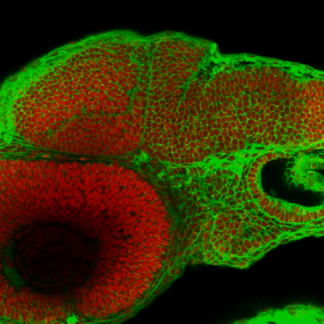
Among vertebrates, fish have an immense capability to regenerate neuronal structures after injuries, offering the chance to study the cellular and molecular basis of neuroregenerative processes.
Besides an acute injury-induced loss of neurons, neurodegenerative diseases lead to a slow but constant progressive loss of neurons, which is irreversible in humans, but may trigger a regenerative response in zebrafish.
In the cerebellum this ability to regenerate neuronal circuits is particularly pronounced.
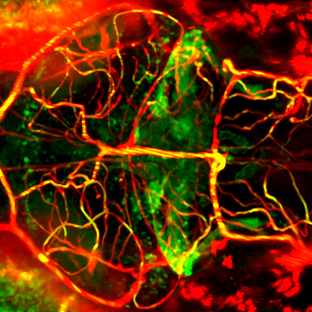
We have established a survival surgery protocol in zebrafish embryos that allows a part or the entire cerebellar anlage to be removed, which is followed by complete cerebellum restoration through regeneration.
Combining zebrafish microsurgery with optical in vivo imaging offers the unique possibility to observe neuroregenerative processes directly in vivo.
This dynamic analysis demonstrates that neuronal migration is crucially involved in larval regeneration inside the cerebellum probably recapitulating the embryonic program of cerebellar development.
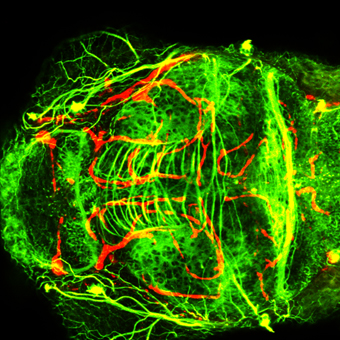
Labeling experiments after induced lesions in the adult zebrafish cerebellum suggest that regeneration in the adult cerebellum also occurs through neuronal migration strikingly similar to the embryonic pattern.
This hints at the exciting possibility of some means of communication between the lesion site and stem cell populations close to the brain ventricle with replacement neurons being guided to their target in a recapitulation of the embryonic program.
Facilitating such reciprocal communication between the injured site and implanted neural stem cells have been reported to significantly increase the repopulation success of lesioned brain areas in mammals. However, the dynamic cellular processes and molecules involved in this communication or promoting regeneration remain to be discovered.
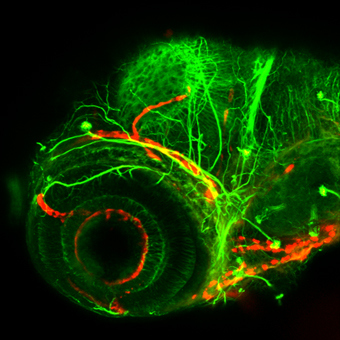
Thus, molecules identified to direct cerebellar neuronal precursors to their target location in the cerebellum during embryogenesis could well play a similar role in regeneration processes at larval and adult stages.
We will test this idea directly in vivo making use of the established interdisciplinary set of techniques bridging imaging, genetics, molecular biology and in vivo microsurgery in zebrafish.
Due to the conservation of the cerebellum among vertebrates findings in zebrafish could well hold true for mammals and humans, reapplied to them and thus aid in developing repopulation strategies for neurodegenerative diseases.
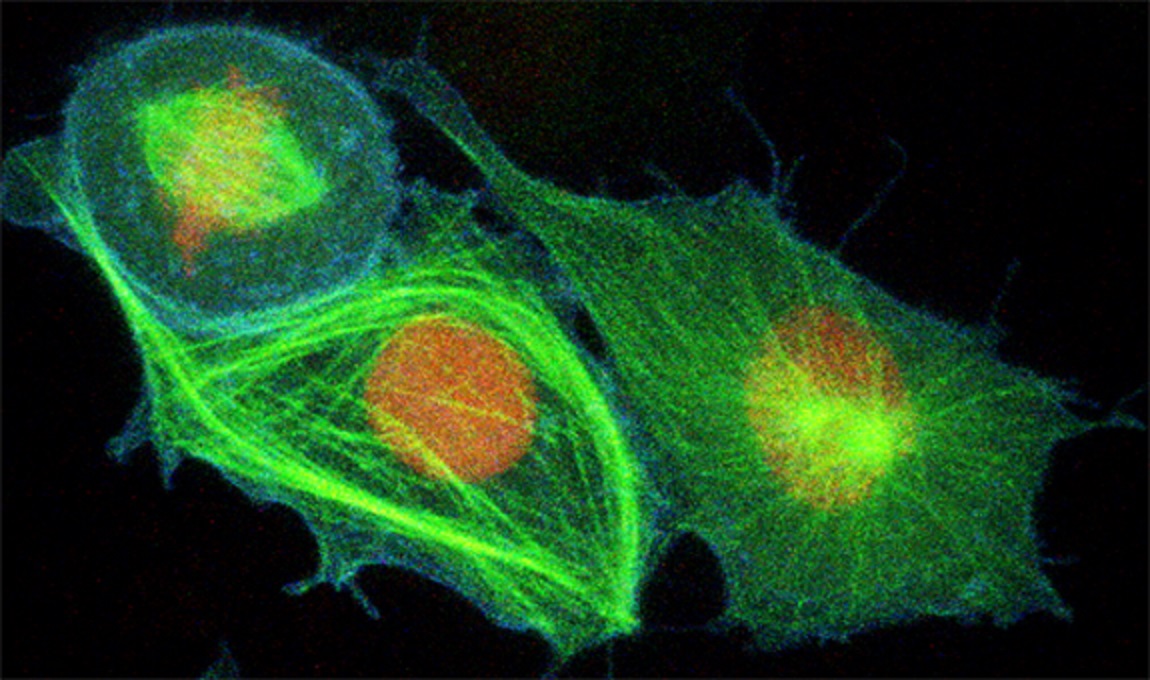

The new ATTACTM approach allows to induce cell type specific ablation. The design of the PC-ATTACTM system permits to specifically trigger the ablation of the Purkinje cell population [Weber, Namikawa et al. 2016], which represents a valuable tool for regeneration studies.

The current research is generously funded by Marie Curie Actions of the European Commission:
Selected publications:
Köster, R. W, Fraser, S. E. (2006). FGF-signaling mediates regeneration of the differentiating zebrafish cerebellum through repatterning of the anterior hindbrain and reinitiation of neuronal migration. Journal of Neuroscience 26: 7293-7304.
Chu, Y., Senghaas, N., Köster, R. W., Wurst, W., Kühn, R. (2008). Novel Caspase-suicide proteins for tamoxifen-inducible apoptosis. Genesis 46: 530-536.
Weber, T., Namikawa, K., Winter, B., Müller-Brown, K., Kühn, R., Wurst, W., Köster, R. W. (2016). Caspase-mediated apoptosis induction in zebrafish cerebellar Purkinje neurons. Development 143: 4279-4287.