Sponsor: DFG
Contact person: Karl Vorländer
Summary of the overall project:
The investigation of the influence of formulating and processing microorganisms on their survival is of particular interest in the context of developing probiotic dosage forms. Probiotic microorganisms interact with the human microbiome and have a positive effect on the health of the patient. For this, the viability of the administered microorganisms is crucial. Different stresses occur during the manufacturing process, caused by temperature and concentration changes as well as mechanical stress. The production of dry dosage forms such as powders or tablets is preferred to liquid or semi-solid products due to their better shelf life and acceptance. However, the drying of microorganisms is usually accompanied by a considerable reduction of the cell viability. Different methods are developed and used to characterize cell viability in order to determine the damage mechanisms in different drying processes and subsequent process steps. Systematic investigations of the influence of process steps as well as process and formulation parameters on the survival of microorganisms are used to scientifically elucidate and rationally design the entire process chain from cell cultivation to packaging of the final dosage form.
Goals and tasks of iPAT
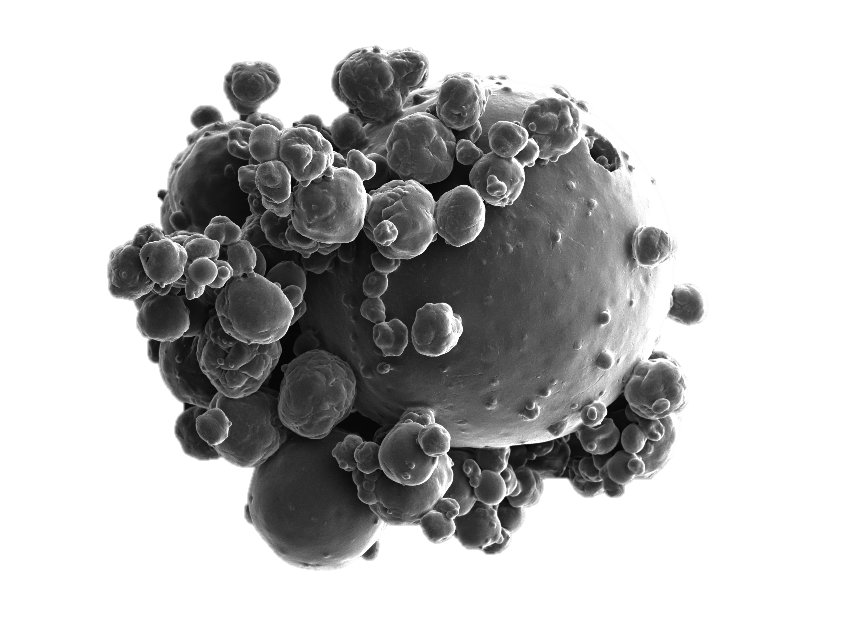
Figure: Spray dried yeast cells